Home » Projects » Shellfish Aquaculture Production and Management Projects » Impact of Temperature Acclimation on Vibrio vulnificus Content for Florida Cultured Clams During Summer Harvest
Home » Projects » Shellfish Aquaculture Production and Management Projects » Impact of Temperature Acclimation on Vibrio vulnificus Content for Florida Cultured Clams During Summer Harvest

Previous laboratory studies conducted during 1996 – 1997 at the UF Aquatic Food Products Lab demonstrated that a gradual or stepwise tempering of hard clams at 68ºF (20ºC) for 12 to 24 hours prior to placement in 45ºF refrigeration acclimatized clams to the decreased temperature and extended the shelf life. The dry tempering method not only enhanced the survival of live clams in customary storage but also kept minimal the microbial consequences. These results warranted both commercial and regulatory consideration. Unfortunately, the recommended tempering scheme violated current harvest regulations.
The harvesting, processing, and distribution of molluscan shellfish is done so in accordance with federal oversight from the U.S. Food and Drug Administration (FDA). Federal regulatory response has included dependence on the established model ordinance of the National Shellfish Sanitation Program as developed through the Interstate Shellfish Sanitation Conference (ISSC) program. The ISSC has a formal issue submission process to accept or reject propositions to modify the Shellfish Sanitation Model Ordinance. A joint submission was made to the ISSC by UF, the FL Department of Environmental Protection (FDEP), the FL Department of Agriculture and Consumer Services (FDACS), and industry at the ISSC annual meeting in 1998. The findings by the ISSC’s Task Force II showed that the model ordinance already reflected an allowance for the state regulatory body to approve other measures proposed by industry to provide controls equivalent to the time/temperature matrix harvest. Consequently, several shellfish wholesalers formally petitioned the state, at that time the FDEP, requesting for a change in the state’s shellfish code. Public workshops were, but the process was delayed when the responsibilities of the state shellfish regulatory authority were transferred to the FDACS by the state legislation in 1999. It was not until August 2000 that the dry tempering method was identified as an alternative process for cultured clams by FDACS and was included in the Comprehensive Shellfish Control Code, Chapter 5L-1, F.A.C. To be approved to include dry tempering into the processing regime, shellfish wholesalers in Florida must apply to the state agency and be approved. Many of the state’s 50 certified shellfish plants that handle cultured clams have done so. During the summer of 2000, validation of the tempering process was conducted by several of the investigators, in conjunction with the FDACS Division of Aquaculture staff, at commercial processing plants. Tempered and non-tempered product samples were collected from June through September for microbiological analysis. These studies demonstrated that tempering did not increase the microbial load of the product as determined by fecal coliform content, standard aerobic plate count, and enumeration of Vibrio-like colonies on selective agar. Further, the total plate and fecal coliform counts never exceeded the established wholesale market standards, and the Vibrio-like organisms did not exceed the initial, limited concentrations measured in the clams after harvest from approved shellfish harvesting waters.
The purpose of this study was to continue to evaluate microbiological consequences during harvesting, processing, tempering, and storage of Florida aquacultured clams. The primary area of concern in this study involved the impact of tempering on possible growth of pathogenic bacteria, particularly Vibrio vulnificus in cultured clams harvested during the summer months. This organism thrives in warm coastal waters approved for shellfish harvesting. The specific objectives were to:1) evaluate the microbial consequences of Vibrio vulnificus before and during tempering and after 24 hours of refrigerated storage; and, 2) make recommendations to direct appropriate commercial response by the Florida clam industry to increase the survival of cultured clams during the warmer months and also offer safe and high quality clams for retailers, restaurants, and consumers.
The verification trials were based on cultured clams harvested from approved waters in Cedar Key, Florida during July, August, and September of 2002. Clam samples were collected during harvesting, or 0 hours. After 6 hours at ambient conditions, one set of clams was placed in a 68ºF environment to temper for 10 hours prior to placement in a cooler at a temperature of 45ºF. Another set of clams from the same lot was held for 10 hours at ambient conditions (the maximum allowed under the time/temperature matrix for summer months) prior to direct placement in refrigerated storage (referred to as No Temper in figure). Three independent trials were examined with duplicate samples for each trial. V. vulnificus was monitored using DNA probe and colony hybridization method as described in the FDA Bacteriological Analytical Manual. This probe is derived for the cytolysin gene, is species-specific, and will theoretically detect as few as 10 to 102 CFU g-1 of clam meat.
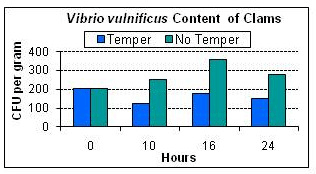 The effects of tempering on V. vulnificus contamination during acclimation of clam shellstock prior to cold storage were examined over time. Continuous monitoring indicated that ambient temperature ranged from 78-86º, and stated incubation temperatures were ±2ºF. Initial levels of V. vulnificus in clams ranged from 50 to 400 CFU g-1 for the three sampling periods with a mean of 203 CFU g-1. During incubation, the highest level of V. vulnificus observed was 790 CFU g-1, which was recorded in the control group that was refrigerated without tempering. After treatment, the mean value for V. vulnificus in clams from the group receiving tempering did not exceed the mean initial value and were <200 CFU g-1 in comparison to the control which was 277 CFU g-1. No significant differences in levels of V. vulnificus were detected in clams from these temperature treatments at 0, 10, 16, or 24 hours. Overall, bacterial levels were notably low and a magnitude (100 times) below those typically obtained from oysters. These data strongly indicate that tempering of clams at 68ºF prior to refrigeration does not promote the growth of V. vulnificus. Further, numbers obtained for tempered samples at 24h incubation were actually lower than refrigerated controls, perhaps due to decreasing the stress on live product as a result of tempering, which has previously been shown to increase shelf life. These results support the use of a step-wise temperature adaptation for the post-harvest treatment of clam aquaculture. Further, these results can be used by industry to verify that tempered clam product is as safe as non-tempered product and does not pose a health risk.
The effects of tempering on V. vulnificus contamination during acclimation of clam shellstock prior to cold storage were examined over time. Continuous monitoring indicated that ambient temperature ranged from 78-86º, and stated incubation temperatures were ±2ºF. Initial levels of V. vulnificus in clams ranged from 50 to 400 CFU g-1 for the three sampling periods with a mean of 203 CFU g-1. During incubation, the highest level of V. vulnificus observed was 790 CFU g-1, which was recorded in the control group that was refrigerated without tempering. After treatment, the mean value for V. vulnificus in clams from the group receiving tempering did not exceed the mean initial value and were <200 CFU g-1 in comparison to the control which was 277 CFU g-1. No significant differences in levels of V. vulnificus were detected in clams from these temperature treatments at 0, 10, 16, or 24 hours. Overall, bacterial levels were notably low and a magnitude (100 times) below those typically obtained from oysters. These data strongly indicate that tempering of clams at 68ºF prior to refrigeration does not promote the growth of V. vulnificus. Further, numbers obtained for tempered samples at 24h incubation were actually lower than refrigerated controls, perhaps due to decreasing the stress on live product as a result of tempering, which has previously been shown to increase shelf life. These results support the use of a step-wise temperature adaptation for the post-harvest treatment of clam aquaculture. Further, these results can be used by industry to verify that tempered clam product is as safe as non-tempered product and does not pose a health risk.
A final report on project results can be viewed below in PDF format.